The immune system is very complicated, but now, it’s on a chip

To quote veteran science writer Ed Yong’s basic nonetheless particularly precise words and phrases in The Atlantic, “The immune system is pretty sophisticated.” As the COVID-19 pandemic experienced made abundantly distinct, science however isn’t going to entirely fully grasp the innovative defense mechanisms that secure us from microbe invaders. Why do some individuals display no signs and symptoms when contaminated with SARS-CoV-2 when other folks put up with from severe fevers and overall body aches? Why do some succumb to cytokine storms of the body’s very own producing? We nonetheless lack actual answers to these thoughts.
Present day experts, however, now have a new tool to assistance them tease out the immune system’s mysteries, many thanks to a group of researchers at the Wyss Institute for Biologically Encouraged Engineering at Harvard University. They cultured human B and T cells inside a microfluidic Organ Chip device and coaxed them to spontaneously form useful lymphoid follicles—structures that reside in lymph nodes and other areas of the human overall body that mediate immune responses. They consist of distinct chambers that harbor “naïve” B cells and T cells, which collectively initiate the cascade of activities that prospects to a complete immune reaction when they are uncovered to a certain antigen.
In addition to enabling researchers to probe the normal purpose of the immune method, these Lymphoid Follicle (LF) Chips can also be employed to forecast immune responses to different vaccines and enable pick out the very best performers, providing considerable improvement in excess of present preclinical products like cells in a dish and non-human primates. The achievement is described these days in Superior Science.
“Animals have been the gold-normal investigate versions for developing and screening new vaccines, but their immune methods vary significantly from our have and do not properly predict how individuals will reply to them. Our LF Chip presents a way to design the complicated choreography of human immune responses to an infection and vaccination, and could significantly speed up the speed and good quality of vaccine development in the upcoming,” explained first author Girija Goyal, Ph.D., a senior workers scientist at the Wyss Institute.
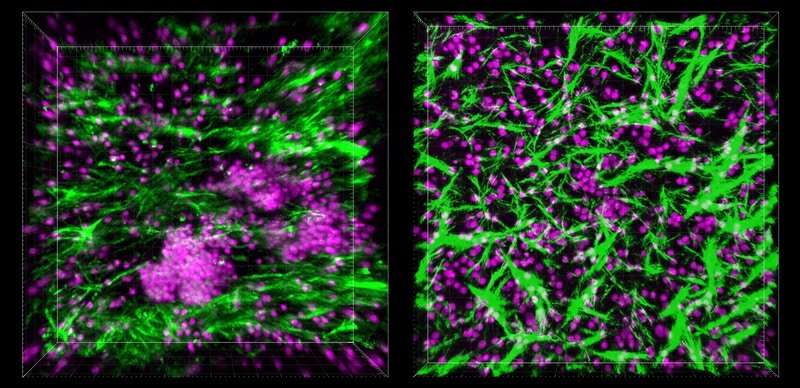
An accidental discovery
Like a lot of excellent scientific discoveries, the LF Chip job is the outcome of serendipity in the lab. Goyal and other Wyss Institute researchers wanted to examine how B and T cells circulating in the blood would adjust their habits the moment they entered a tissue, so they obtained these cells from human blood samples and cultured them inside of a microfluidic Organ Chip product to replicate the actual physical ailments they would expertise when they encountered an organ.
When the cells were put inside 1 of the two channels inside of the gadget, almost nothing extraordinary happened—but when the researchers commenced the circulation of tradition medium as a result of the other channel to feed the cells, they have been surprised to see that within the Organ Chip, the B and T cells begun to spontaneously self-arrange into 3D buildings that appeared related to “germinal facilities”—structures within just LFs where by intricate immune reactions consider location. “It was so unpredicted that we entirely pivoted from the initial experiment and focused on attempting to determine out what they were being,” mentioned Goyal.
When the scientists began probing the mysterious buildings that had formed within the Organ Chip less than flow ailments, they uncovered that the cells had been secreting a chemical termed CXCL13. CXCL13 is a hallmark of LF development, both inside lymph nodes and in other sections of the human body, in reaction to serious irritation, this kind of as in cancer and autoimmune situations.
The group also identified that B cells inside the LFs that self-assembled on-chip also expressed an enzyme named activation-induced cytidine deaminase (Aid), which is vital for activating B cells versus unique antigens and is not current in B cells that are circulating in the blood.
Neither CXCL13 nor Support have been existing in cells that were being cultured in a common 2D dish, suggesting that the researchers had without a doubt correctly produced functional LFs from circulating blood cells.
In LFs in the human overall body, activated B cells experienced and differentiate into many sorts of progeny cells which includes plasma cells, which secrete significant amounts of antibodies versus a certain pathogen. The team detected the existence of plasma cells in the LF Chips soon after they used quite a few stimuli utilised in the laboratory to activate B cells, such as the mix of the cytokine IL-4 and an anti-CD40 antibody, or dead bacteria. Remarkably, the plasma cells have been concentrated in clusters inside of the LFs, as they would be in vivo.
“These findings were being specifically thrilling for the reason that they confirmed that we experienced a purposeful design that could be utilized to unravel some of the complexities of the human immune method, such as its responses to many styles of pathogens,” reported Pranav Prabhala, a Technician at the Wyss Institute and 2nd author of the paper.
Predicting vaccine efficacy on-a-chip
Now that the researchers experienced a purposeful LF model that could initiate an immune reaction, they explored irrespective of whether their LF Chip could be used to replicate and review the human immune system’s reaction to vaccines.
In the human system, vaccination induces particular cells termed dendritic cells to acquire up the injected pathogen and migrate to lymph nodes, exactly where they existing fragments of them on their surface area. There, these antigen-presenting cells activate the B cells with the aid of regional T cells in the LF, creating the B cells to differentiate into plasma cells that generate antibodies versus the pathogen. To replicate this procedure, the researchers additional dendritic cells to LF Chips together with B and T cells from four individual human donors. They then inoculated the chips with a vaccine versus the H5N1 strain of influenza together with an adjuvant named SWE that is recognised to improve immune responses to the vaccine.
LF Chips that received the vaccine and the adjuvant developed noticeably far more plasma cells and anti-influenza antibodies than B and T cells grown in 2D cultures or LF Chips that acquired the vaccine but not the adjuvant.
The workforce then repeated the experiment with cells from eight distinct donors, this time utilizing the commercially accessible FluzoneⓇ influenza vaccine, which guards towards three distinct strains of the virus in people. When once more, plasma cells and anti-influenza antibodies were being existing in sizeable figures in the dealt with LF Chips. They also measured the degrees of 4 cytokines in the vaccinated LF Chips that are identified to be secreted by activated immune cells, and identified that the degrees of 3 of them (IFN-γ, IL-10, and IL-2) were identical to those discovered in the serum of humans who experienced been vaccinated with FluzoneⓇ.
The Wyss researchers are now making use of their LF Chips to test a variety of vaccines and adjuvants in collaboration with pharmaceutical providers and the Gates Basis.
“The flurry of vaccine advancement attempts sparked by the COVID-19 pandemic were spectacular for their pace, but the enhanced need all of a sudden created traditional animal models scarce resources. The LF Chip gives a less expensive, more quickly, and additional predictive model for studying human immune responses to both equally bacterial infections and vaccines, and we hope it will streamline and make improvements to vaccine progress towards a lot of health conditions in the potential,” reported corresponding author Donald Ingber, M.D., Ph.D., who is the Founding Director of the Wyss Institute as nicely as the Judah Folkman Professor of Vascular Biology at Harvard Health care School (HMS) and Boston Children’s Medical center, and Professor of Bioengineering at the Harvard John A. Paulson College of Engineering and Utilized Sciences.
A new device for learning COVID’s influence on intestine health
Girija Goyal et al, Ectopic Lymphoid Follicle Development and Human Seasonal Influenza Vaccination Responses Recapitulated in an Organ‐on‐a‐Chip, Sophisticated Science (2022). DOI: 10.1002/advs.202103241
Citation:
The immune program is extremely difficult, but now, it is really on a chip (2022, March 15)
retrieved 16 March 2022
from https://medicalxpress.com/information/2022-03-immune-complex-chip.html
This document is subject to copyright. Apart from any reasonable working for the reason of personal research or research, no
component may possibly be reproduced with no the penned authorization. The written content is presented for data applications only.







